titanatitanium calcium oxide
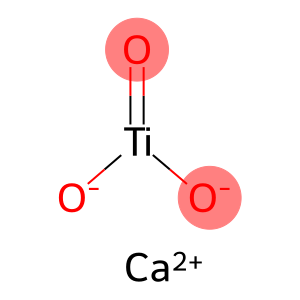
Calcium titanate
CAS: 12049-50-2
Molecular Formula: CaO3Ti
titanatitanium calcium oxide - Names and Identifiers
| Name | Calcium titanate |
| Synonyms | ct calcium titanate calcium Calcium titanate oxygen(-2) anion calcium metatitanate titanium(+4) cation Calciumtitaniumtrioxide calcium titanium trioxide titanatitanium calcium oxide dicalcium tetraoxidotitanium |
| CAS | 12049-50-2 |
| EINECS | 234-988-1 |
| InChI | InChI=1/2Ca.4O.Ti/q2*+2;4*-1;/r2Ca.O4Ti/c;;1-5(2,3)4/q2*+2;-4 |
titanatitanium calcium oxide - Physico-chemical Properties
| Molecular Formula | CaO3Ti |
| Molar Mass | 135.94 |
| Density | 4.1 g/mL at 25 °C (lit.) |
| Melting Point | 1975°C |
| Water Solubility | Insoluble in water. Soluble in oirganic and inorganic solution. |
| Appearance | White to Brown Powder |
| Specific Gravity | 4.1 |
| Color | Beige |
| Storage Condition | Room Temprature |
| MDL | MFCD00015981 |
| Use | Calcium titanate is a basic inorganic dielectric material with excellent dielectric properties, temperature properties, mechanical properties and optical properties. It is widely used in ceramic capacitors, PTC thermistors, microwave antennas, filters, stainless steel electrodes and other fields. |
titanatitanium calcium oxide - Risk and Safety
| WGK Germany | 2 |
| RTECS | XR1468666 |
| TSCA | Yes |
titanatitanium calcium oxide - Nature
Open Data Verified Data
crystalline powder. Cubic crystal system. Relative density 3. 98; Melting point 1980 ℃. High thermal decomposition of toxic calcium and titanium smoke.
Last Update:2024-01-02 23:10:35
titanatitanium calcium oxide - Preparation Method
Open Data Verified Data
with titanium oxide and calcium oxide as raw materials, the amount of calcium oxide, titanium oxide and other additives are fully mixed, after grinding, extrusion molding, and then calcined at 1350, after calcination, the product was pulverized and ground.
Last Update:2022-01-01 09:05:38
titanatitanium calcium oxide - Use
Open Data Verified Data
for the production of medium and high voltage ceramic capacitors, PTC thermistors and other precision electronic components. Pure calcium titanate is difficult to sinter into ceramic, and 1% ~ 2% zirconia can be added.
Last Update:2022-01-01 09:05:38
titanatitanium calcium oxide - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| use | calcium titanate is a basic inorganic dielectric material with excellent dielectric properties, temperature properties, mechanical properties and optical properties, and is widely used in ceramic capacitors, PTC thermistors, microwave antennas, filters, stainless steel electrodes and other fields. Perovskite is the name of calcium titanate mineral. The structure of perovskite involves many inorganic crystalline materials. In-depth understanding of the structure and changes of perovskite will play an important role in the research and development of inorganic functional materials. Calcium titanate (CaTiO3) is a cubic crystal system. Its structure can be regarded as small Ti4 at the apex of the cubic unit cell, large Ca2 + at the center of the unit cell, and O2-at the center of the prism of the unit cell. There are 6 O2-coordination around each Ti4, which is regular octahedral, and 12 O2-coordination around each Ca2, which is cubic octahedral (A type). The cubic octahedron has 24 edges, 12 vertices, and 14 faces, of which 14 faces are six squares and eight regular triangles. If the origin of the unit cell is moved to Ca2, Ca2 and O2-are arranged in the closest cubic stacking together in an orderly manner to form a face-centered cubic unit cell. Ti4 occupies the center point of the unit cell in an orderly manner, I .e. among the 6 octahedral voids composed of O2-, the face-centered cubic unit cell has 4 octahedral voids, and the other 3 octahedral voids in the center of the edge are all composed of 4 O2-and 2 Ca2, these 3 octahedral voids are not occupied by Ti4 (Type B). |
| production method | 1. take titanium oxide and calcium oxide as raw materials, fully mix calcium oxide, titanium oxide and other additives in the same amount, grind and extrude, then calcinate at 1350 ℃, then crush and grind them to obtain the product. 2. Mix TiO2 and CaO in water, add oxalic acid solution under full stirring, then separate, wash, dry and calcine at 925 ℃. 3.TiCl4 reacts with oxalic acid solution to obtain titanium oxalate solution first, and then mix it with CaCl2 solution to obtain calcium titanium oxalate, which is filtered, dried and calcined at 1050 ℃ to generate calcium titanate. 4. React with calcium hydroxide and calcium nitrate mixture with titanium dioxide at 575 ℃. 5. |
| category | toxic substances |
| toxicity classification | low toxicity |
| acute toxicity | oral-rat LD50: > 12000 mg/kg; Abdominal cavity-mouse LD50: 5300 mg/kg |
| flammability hazard characteristics | high heat decomposition discharges toxic calcium and titanium smoke |
| storage and transportation characteristics | warehouse low temperature ventilation and drying |
| fire extinguishing agent | water |
Last Update:2024-04-10 22:29:15
Supplier List
Multiple SpecificationsSpot supply
Product Name: Calcium titanate Visit Supplier Webpage Request for quotationCAS: 12049-50-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Calcium titanate Request for quotation
CAS: 12049-50-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 12049-50-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: CALCIUM TITANATE Visit Supplier Webpage Request for quotationCAS: 12049-50-2
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: CALCIUM TITANATE Request for quotationCAS: 12049-50-2
Tel: 15335994747
Email: 3521670276@qq.com
Mobile: 15335994747
QQ: 3521670276
Wechat: A2020071098
Product Name: CALCIUM TITANATE Request for quotation
CAS: 12049-50-2
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 12049-50-2
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: Calcium titanate Request for quotation
CAS: 12049-50-2
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 12049-50-2
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Product Name: Calcium titanate Visit Supplier Webpage Request for quotation
CAS: 12049-50-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 12049-50-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple SpecificationsSpot supply
Product Name: Calcium titanate Visit Supplier Webpage Request for quotationCAS: 12049-50-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Calcium titanate Request for quotation
CAS: 12049-50-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 12049-50-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: CALCIUM TITANATE Visit Supplier Webpage Request for quotationCAS: 12049-50-2
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: CALCIUM TITANATE Request for quotationCAS: 12049-50-2
Tel: 15335994747
Email: 3521670276@qq.com
Mobile: 15335994747
QQ: 3521670276
Wechat: A2020071098
Product Name: CALCIUM TITANATE Request for quotation
CAS: 12049-50-2
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 12049-50-2
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: Calcium titanate Request for quotation
CAS: 12049-50-2
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 12049-50-2
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Product Name: Calcium titanate Visit Supplier Webpage Request for quotation
CAS: 12049-50-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 12049-50-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History






